(There is some evidence that philosophers in both India and China made similar speculations at about the same time. The earliest significant ideas to survive are from the ancient Greeks in the fifth century BC, especially from the philosophers Leucippus and Democritus. People have long speculated about the structure of matter and the existence of atoms. It has dimensions of reciprocal mol and its value is equal to $6.02214129 \cdot 10^ 1 0 − 10 m, or 100 pm).
electromagnetic force: a long-range fundamental force that acts between charged bodies, mediated by the exchange of photons.Dalton established his atomic theory based on the fact that the masses of reactants in specific chemical reactions always have a particular mass ratio.Scattered knowledge discovered by alchemists over the Middle Ages contributed to the discovery of atoms.The atom is a basic unit of matter that consists of a dense central nucleus surrounded by a cloud of negatively charged electrons.This was the first reported nuclear reaction, One hydrogen nucleus was knocked off by the impact of the alpha particle, producing oxygen-17 in the process. Rutherford determined that the only possible source of this hydrogen was the nitrogen, and therefore nitrogen must contain hydrogen nuclei. After experimentation Rutherford traced the reaction to the nitrogen in air, and found that the effect was larger when alphas were produced into pure nitrogen gas. These experiments began when Rutherford noticed that when alpha particles were shot into air (mostly nitrogen), his scintillation detectors displayed the signatures of typical hydrogen nuclei as a product. Earlier, Rutherford learned to create hydrogen nuclei as a type of radiation produced as a yield of the impact of alpha particles on hydrogen gas these nuclei were recognized by their unique penetration signature in air and their appearance in scintillation detectors. In 1917 (in experiments reported in 1919), Rutherford proved that the hydrogen nucleus is present in other nuclei, a result usually described as the discovery of the proton. )Įlectron Beam: A beam of electrons deflected in a circle by a magnetic field. (The hydrogen-1 atom, however, has no neutrons, and a positive hydrogen ion has no electrons. The constituent particles of an atom (each discovered independently) are: the electron, the proton and the neutron. Though originally viewed as a particle that cannot be cut into smaller particles, modern scientific usage denotes the atom as composed of various subatomic particles. cathode: An electrode through which electric current flows out of a polarized electrical device.alpha particle: A positively charged nucleus of a helium-4 atom (consisting of two protons and two neutrons), emitted as a consequence of radioactivity.scintillation: A flash of light produced in a transparent material by the passage of a particle.These particles, later called neutrons, had a similar mass of the protons but did not have the same characteristics as protons. In 1932, James Chadwick showed that there were uncharged particles in the radiation he was using.Rutherford proved that the hydrogen nucleus is present in other nuclei.

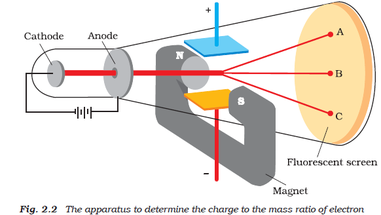
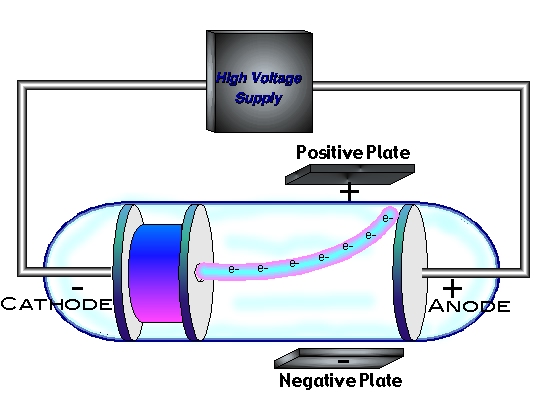
Thomson performed experiments studying cathode rays and discovered that they were unique particles, later named electrons.
